The Need
Clinicubes is a specialized software product developed by BGO Software of the Clinical Research Industry.
It aims to facilitate the way professionals conduct the whole project lifecycle in the clinical research process. Being a clinical trial management system – CTMS, it delivers a number of advantages in the form of integrated solutions, which provide aid in every aspect of the clinical research process. Clinicubes simplifies the complexity behind many processes and tasks, including planning, performing and reporting.
It also accommodates the needs of research organizations to collect, retain, document and store scientific or patient data. The software tracks deadlines, schedules visitations and monitors the whole progress in a fast and easy manner. In order to save time, Clinicubes shortens the prolonged implementation of different operations. It also reduces extra costs and guarantees ultimate end-to-end customer experience.
The Challenge
In order for the product to effectively manage the clinical trial’s pipeline, our SaaS includes functionalities that are at the heart of a successful research project completion.
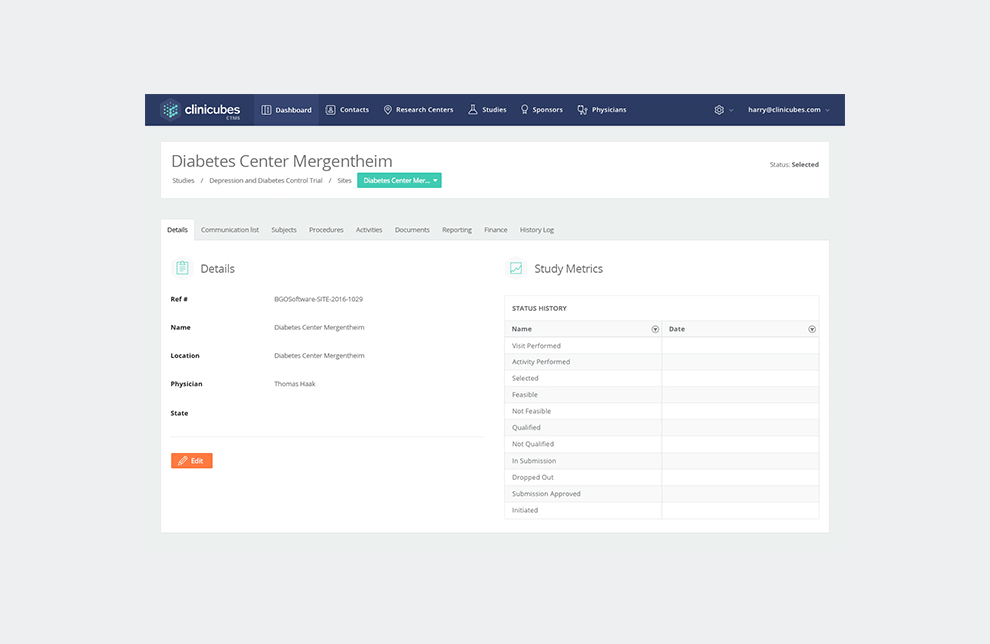
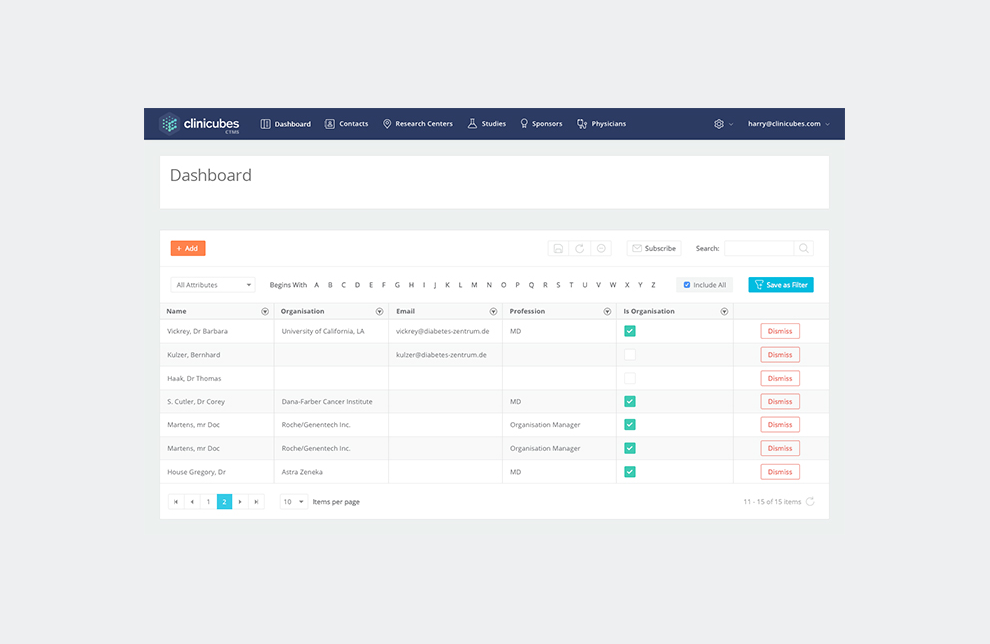
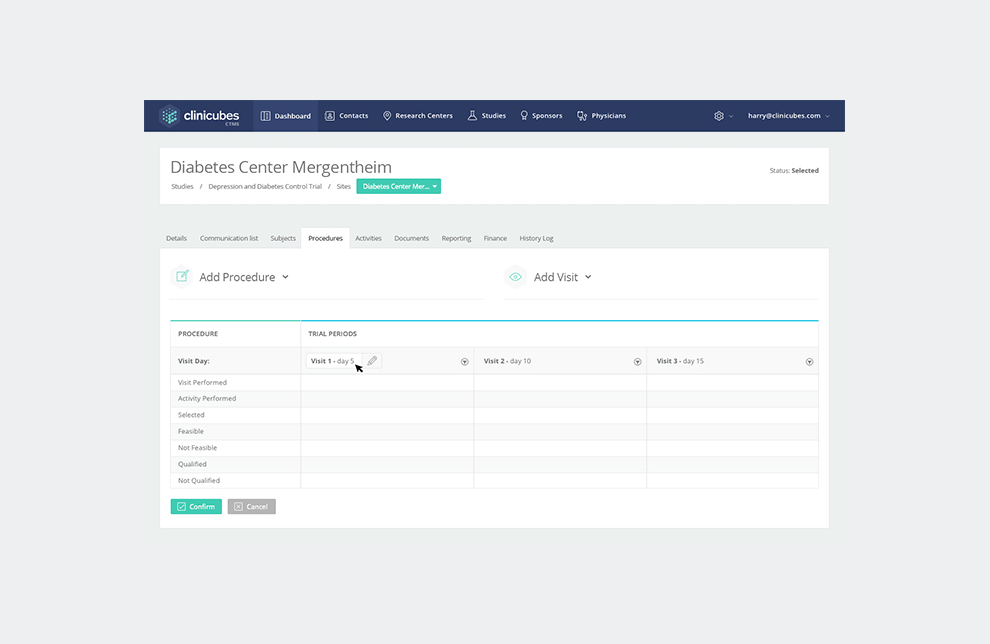
Site tracking and management – The system enables research professionals to monitor sites and regulatory processes by gathering all of the required information for further decision making. Clinicubes facilitates site identification and recruitment. It provides full control and tracking over subject enrollment and subjects database.
Physicians and health service instructions database – Clinicubes allows you to collect, evaluate and analyze data from multiple studies and different locations on a single report. It provides professionals with reporting options of how the trial is being carried out against pre-set criteria and targets. What is more, it enables users to archive study documents and communicate study performance data as well as other operational reports through interactive dashboards.
Overall project tracking and collaboration tools – Our clinical trial management system offers plenty of tools for project tracking and team collaboration. It helps you schedule key activities or plan important events. You will also find dynamic dashboards with key metrics, various options for alerts and reporting, calendar scheduling features, email reports, and various collaboration tools.
Тhe Solution
Solution
Clinicubes offers the combination of an impressive array of functionalities and easiness of use, which allows users to not only plan but also shape successful research results.
- Overview and management of clinical trials
- Study events and activities management.
- Milestone tracking
- Financial forecasting, budgeting, invoices and payments
- Site management and regulatory process tracking
- Subjects database and study enrollment tracking
- Physicians database and study performance data
- Health service institutions database
- Project documents collection and tracking
- Dynamic dashboards with key metrics
- Alerts and reporting, calendar scheduling, email reports
- Integration with the most popular clinical research applications
- Migration and import of data collected
Value Delivered
BGO’s custom SaaS solution, entirely meets the needs of clinical trial organizations to collect and archive patient or scientific data. The CTMS can be utilized for scheduling visitations, tracking deadlines, managing budget, and monitoring the entire progress. In addition, Clinicubes saves time by minimizing the implementation time for various procedures. It also decreases the additional costs and ensures there is ultimate customer experience.
- Overview of clinical trial objectives, timetables, phases
- Monitor frequency and collect care report forms (CRFs)
- Manage study events, activities and milestone tracking
- Prepare study contracts, track study costs and carry out financial forecasting
- Track key information related to protocol requirements
- Do quick budgeting, manage invoices and billing complaints, set up secure payments.