The Need
BGO Software’s client, a prominent global pharmaceutical company, faced the challenge of managing and analyzing vast amounts of data generated during their manufacturing processes. To address this need, they required a sophisticated tool that could facilitate data analysis and accelerate their decision-making process.
The Challenge
The management and analysis of data in the pharmaceutical industry’s Drug Substance and Drug Product processes presents a significant challenge. With sensors and measurement equipment becoming more accessible, the amount of data generated is growing exponentially. The key to unlocking the full potential of this data lies in converting it into actionable insights through data intelligence. Integration with the historian system is a critical step in this process. Once technically implemented, it is equally essential to establish robust business processes to ensure efficient utilization and maintenance.
Тhe Solution
The solution is a powerful global system that integrates various mathematical approaches to process data evaluation, enabling data-driven decision-making and accelerating future project implementation across the network.
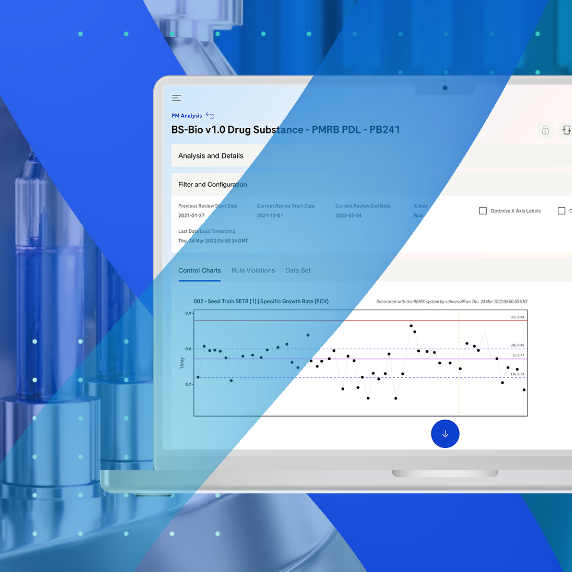
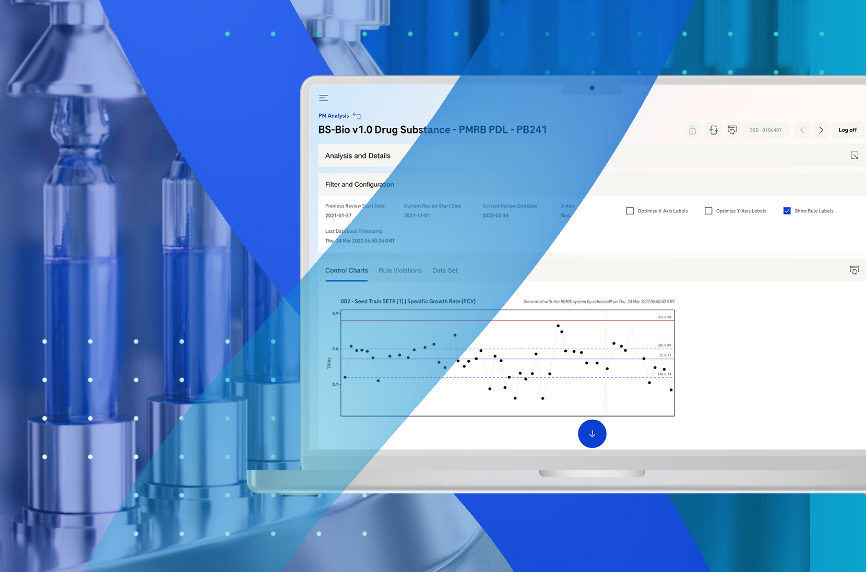
The solution leverages multivariate statistics such as Principal Component Analysis (PCA), Partial Least Squares (PLS), and Artificial Neural Network (ANN), among others, to generate insights. By combining these techniques with regression methods, it is possible to predict the process course or important Key Performance Indicators (KPI), making process monitoring more efficient.
In contrast to traditional univariate approaches, the solution enables the assessment of many multivariate process parameters at once, allowing for a quick assessment of whether the process is still on track. This data-driven analysis enables more informed decision-making.
Additionally, the solution offers a method which is an innovative approach to monitoring column performance. By leveraging in-process data from routine manufacturing operations, typically a step change in conductivity, this method provides a practical and cost-effective way to monitor column integrity throughout its lifetime. Statistical Process Control (SPC) techniques can also be used to identify trends in output parameters, further enhancing the monitoring process.
Value Delivered
The platform’s unique capability lies in its ability to extract process data from various GMP processes and use it to build a non-GMP environment model. This approach enables users to make informed decisions based on their expertise and available GMP data.
Importantly, the platform does not replace or alter existing GMP procedures. Rather, it serves as a complementary tool that provides a diagnosis of process performance to supplement existing GMP procedures.
In practice, the implementation of this platform at manufacturing sites is entirely outside the scope of GMP and does not require a level 1, 2, or 3 change per gspX. The implementation location will determine whether a level 0 change is necessary, or if the platform can be integrated without the need for technical change management processes.