The Need
Pharmaceutical and biotechnology company based in Switzerland needed an integrated solution for the product quality management system, process monitoring and data trending that guarantees that a control state will be maintained and that continuous operation within a control state will be verified. Continuous, real-time monitoring of the entire product and process lifecycle in the production of pharmaceuticals was a must.
The Challenge
The client sought to improve overall performance and agility in current business practices for data collection, data quality/governance, analytics, and reporting so that timely insights could be gained across diverse processes and knowledge could be easily shared cross-functionally throughout the corporate network. At the time the project began, the client was struggling with numerous issues and “gold standard technologies,” including:
- Paper-based documents were manually transcribed and reviewed for compliance prior to analysis, which was very time consuming and labor intensive
- Increasing compliance risk in monitoring products due to more and more data to be analyzed and reported on
- Different approaches to monitoring and reporting, resulting in a multitude of misunderstandings
- Limited data accessibility
Тhe Solution
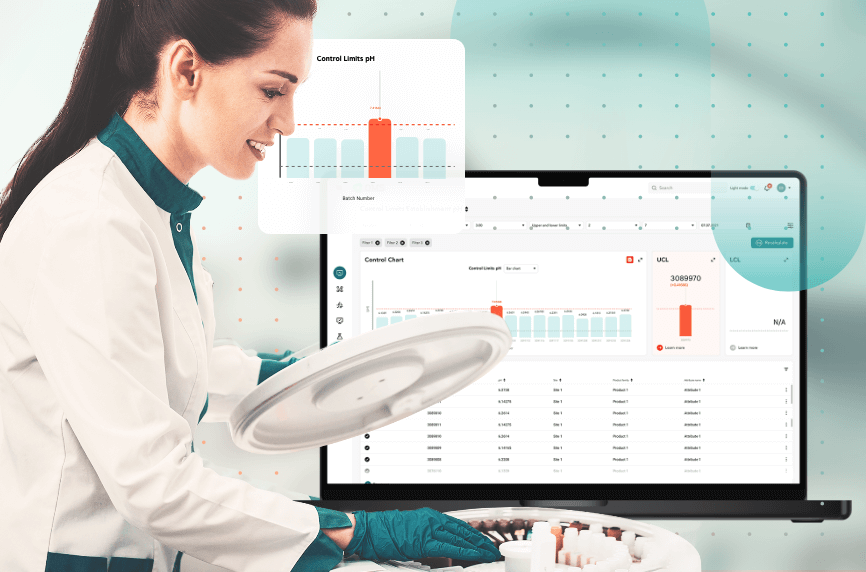
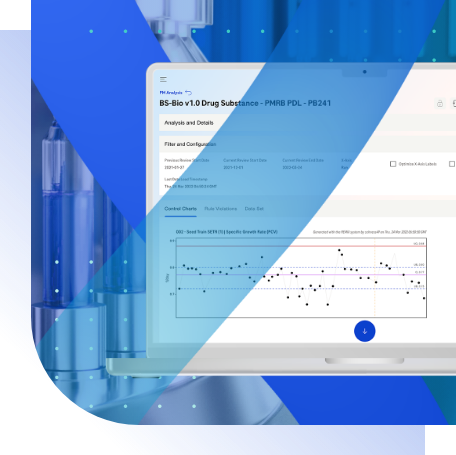
For the customer’s needs, BGO Software is developing a manufacturing intelligence solution that automatically collects, analyses, and reports global manufacturing data. The solution is a web based GMP platform that enables real-time process and product monitoring (PPM) and continuous process verification (CPV) in the manufacturing of pharmaceutical products (drugs). In compliance with FDA 21 CFR Part 11 and GMP, this solution is a guarantee of the highest product quality. Among other features, the system supports workflow review and approval, as well as audit trail capabilities for managing, controlling, and monitoring information. In addition, the system contains user-entered master data so that PPM data from different sources and business processes can be correlated. Finally, various trending and reporting requirements are met by performing statistical analysis and executing KPI calculations.
Key Outcomes of the solution
- Monitor the entire product and process lifecycle in real time while meeting regulatory expectations and improving cGMP compliance
- Proactively identify and reduce variations in manufacturing processes
- Standardization of product manufacturing quality
- Improve reliability of product supply
- Improve business processes to capture process monitoring information and simplify GMP reporting
- Standardization of monitoring approaches worldwide
- Ensuring coordination and quality across manufacturing locations
Value Delivered
The main values delivered are the Process and Product Monitoring & Continued Process Verification Achieved.
The system is complaint with process and product monitoring by:
- Creating attributes and mappings
- Establishing control limits for attributes
- Monitoring variation with control chart
- Performing trend analysis to assess the state of control
- Documenting trends (rule violations) monitoring
The solution which BGO Software has been building is targeted to be a Continued Process Verification (CPV) System. The main capabilities are data to be collected, contextualized, analyzed, and visualized to support regular monitoring and root cause investigations. Thanks to it, the Swiss-based pharmaceutical and biotechnology company ensures that processes remain in a constant ‘state of control’. In this way, they ensure the final product quality after the process has been validated.
The main value delivered to our client client through this solution is the millions of CHF saved each year (exceeding 6 million). In addition to substantial financial savings, the solution eliminates up to 100 hours of manual work and reporting—previously, manual reporting required approximately 130 hours, whereas our automated approach reduces it to just 29 hours.
The solution encompasses more than 3,000 attribute definitions, which had no prior digital storage representation. Furthermore, it manages over a million attribute values, supporting almost 1,000 users who rely on data from more than 50 data connections. This case study underscores the significant financial benefits and operational efficiencies achieved through the adoption of the software solution.