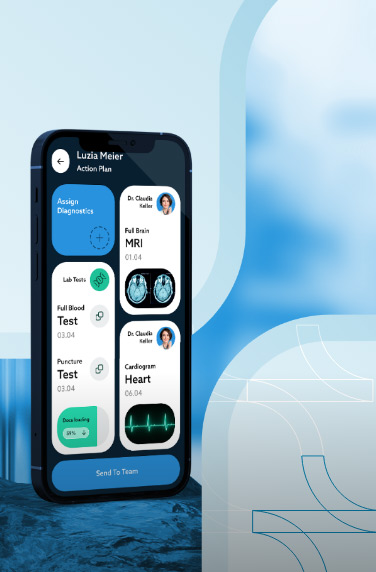
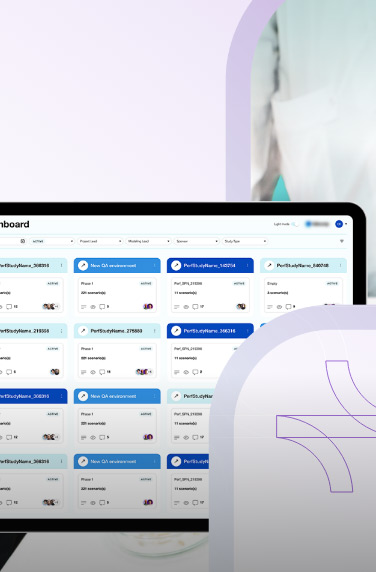
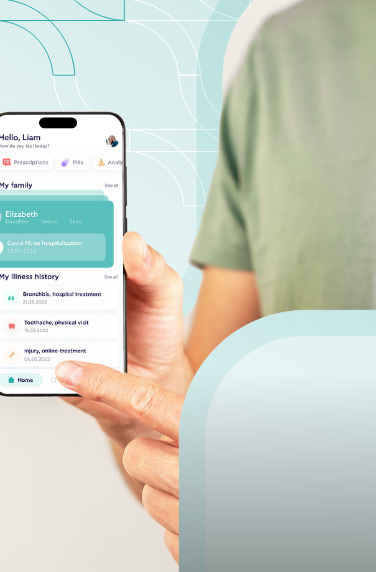
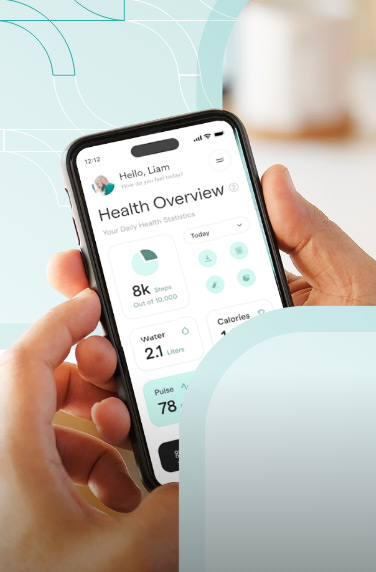
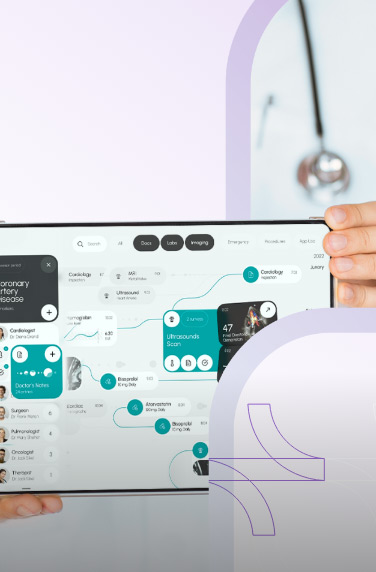
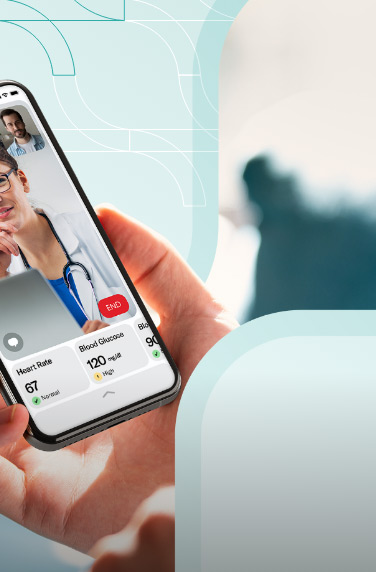
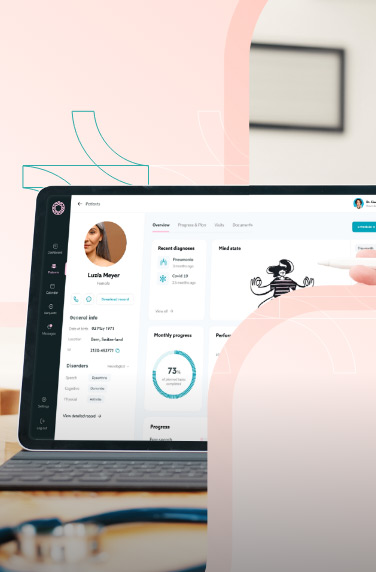
A clinical trials management system assists hospitals, pharmacies, or research sites manage clinical trials. The software system handles patient data, financing, scheduling, reporting, and research operations.
Clinical trial management software also helps with regulatory compliance by keeping the health and financial data of the clinical researchers. Other features can include patient enrollment and recruitment, study planning, etc.
However, a CTMS solution must not be mistaken for an Electronic data capture (EDC). The EDC is focused primarily on collecting patient data, while clinical trials management software focuses on the project management side of clinical research.