The pharmaceutical industry is expanding worldwide, so safety monitoring needs to be consistent. Drug development is getting more complex, and information spreads faster than ever. New technologies also make strong Good Pharmacovigilance Practices (GVP) more important.
In this article, we will go through everything you need to know about GVP in detail, starting with its key principles and aspects. We will mention the regulatory measures and institutions involved, the compliance and quality assurance, and the role of the marketing authorization holder.
Definition of good pharmacovigilance practice (GVP)

Good Pharmacovigilance Practice (GVP) is a set of guidelines that help track and report adverse drug reactions (ADRs) and other safety concerns related to medicines.
The primary goal of pharmacovigilance is to enhance patient safety by identifying and minimizing the risks associated with the use of drugs.
Consistent safety standards help protect patients worldwide by ensuring strong monitoring systems. They also encourage ongoing improvements in drug safety and strengthen public trust in medicines.
In addition, the World Health Organization (WHO) defines pharmacovigilance as:
“The science and activities relating to the detection, assessment, understanding, and prevention of adverse effects or any other drug-related problem.”
The organization specifies that any newly developed drug must meet three criteria prior to approval – good quality, effectiveness, and safety for intended purposes.
The WHO includes the following key aspects in GVP:
- Interactions of medicines;
- Abuse and misuse of medicines;
- Counterfeit medicines;
- Medication errors;
- Adverse drug reactions and events;
- Lack of efficacy.
The competence of pharmacovigilance practice not only includes medicines but also vaccines, blood products, herbals, medical devices, biological, and traditional/complementary substances.
6 key principles of GVP
According to the European Medicines Agency (EMA) and its guidelines of GVP, in establishing and maintaining a robust pharmacovigilance system, several key principles must be upheld:
- Patient-centric focus. The safety needs of patients, healthcare professionals, and the general public concerning medicines should be prioritized and addressed effectively.
- Leadership and motivation. Strong leadership is key to implementing a quality system. Upper management should not only guide the process but also inspire employees to meet quality goals. Their active involvement helps keep everyone motivated and committed.
- Structured resources and tasks. Resources and tasks must be organized in a structured manner to facilitate proactive, risk-proportionate, continuous, and integrated pharmacovigilance. This is crucial for effective risk management.
- Comprehensive evidence evaluation. Gathering all available evidence on a medicine’s risks and benefits is crucial. Decisions should consider every factor that might affect its safety and effectiveness.
- Collaborative partnerships. Strong cooperation between stakeholders—such as drug manufacturers, health authorities, public health organizations, and healthcare professionals—is essential. Working together within legal guidelines helps improve drug safety and protect patients.
By following these principles, organizations can establish a proactive, patient-centered, and collaborative pharmacovigilance approach, ensuring the continual improvement of drug safety practices.
What are the key aspects of good pharmacovigilance practice?
Aside from the key basic principles, some other more detailed vital aspects should be included in every GVP.
Adverse drug reaction reporting
Companies involved in the development, manufacturing, and marketing of pharmaceuticals are required to establish systems for the collection, assessment, and reporting of adverse reactions related to their products.
To ensure smooth reporting, healthcare professionals or drug manufacturers must report any suspected adverse reactions to a national pharmacovigilance center. This helps track and manage potential risks linked to medicines.
An adverse drug reaction report must contain the following elements:
- An identifiable healthcare professional;
- An identifiable patient;
- At least one suspected substance or medicinal product;
- At least one suspected reaction.
Reports are made in writing (e.g. using report forms), by telephone, electronically, or in any other approved way.
Discover how we can help outsource Healthcare projects efficiently
Risk management
GVP emphasizes the importance of assessing and managing risks associated with drug products throughout their lifecycle. This involves continuously monitoring and updating risk-benefit profiles based on new information.
The benefit to risk can be improved either by increasing the benefits, or by reducing the risks by minimizing risk factors.
This could happen, for example, by ensuring the following:
- Contra-indicating the use in patients, particularly at risk;
- Lowering the dosage of the new drugs;
- Introducing pre-treatment tests to identify patients at risk;
- Monitoring during treatment for early diagnosis of hazards that are reversible.
When suggesting ways to improve a drug’s benefit-risk balance, it’s important to consider how practical these measures are in real-world use. For example, limiting the drug to patients who would benefit most or when no alternatives exist should be feasible in everyday medical practice.
Signal detection
Signal detection is the process of identifying possible safety concerns or new risks related to a drug. It involves analyzing data from different sources to spot patterns or trends that could signal a safety issue.
If anything is detected, reports are collected and validated by the pharmacovigilance center and are usually entered into a database.
Serious reactions should be handled with the highest priority. The database is used to identify potential signals and analyze data to clarify risk factors, apparent changes in reporting profiles, etc.
Documentation and record-keeping
Maintaining detailed and accurate records of pharmacovigilance activities is crucial. This includes documentation of adverse event reports, risk assessments, and any actions taken to address safety concerns.
Before the study commences a protocol must be finalized that explains the following:
- The aims and objectives of the study;
- The methods to be used (including statistical analysis and justification of sample size);
- Record-keeping that is to be maintained.
The marketing authorization holder should submit the protocol plus any proposed communications to doctors to the relevant regulatory authorities at least one month before the planned start of the study.
The authorities may comment as necessary. The responsibility for the conduct of the study will, however, rest with the sponsoring pharmaceutical company.
Communication
Effective communication of safety information is vital to ensure that healthcare professionals, regulatory authorities, and patients are informed about potential risks associated with a drug. This includes the timely dissemination of safety alerts and updates.
A clear example of a communication process is when a drug manufacturer informs regulatory authorities when a study begins. They typically provide progress reports every six months or as required, ensuring proper record-keeping and transparency.
GVP emphasizes the establishment of a quality management system to ensure that pharmacovigilance activities are conducted in a systematic, consistent, and compliant manner.
We will go through QMS in detail further in the article.
Data collection and analysis in GVP

Understanding how to collect data for GVP and ensuring the ability to perform all necessary audit trails is crucial for effective performance. Maintaining the conformity of stored data with the initial reports is vital.
This can be achieved through a quality control process that includes validating the stored data against the original data or its images.
The storage system must ensure that all data can be traced. It should track when and where data was received, any modifications made, and when and where it was shared. This helps maintain a complete audit trail.
In the realm of pharmacovigilance, adverse event reporting is a critical component and can be categorized as either spontaneous or expedited:
- Spontaneous reports, sourced from healthcare professionals, patients, and consumers, provide valuable insights into potential side effects associated with medicinal products.
- Expedited reporting is mandated for serious and unexpected adverse events, requiring swift communication with regulatory authorities.
To monitor these events effectively, structured methods are used to collect safety data. This includes standardized details such as patient demographics, medical history, other medications taken, and a full description of the adverse event.
Advanced information systems can improve this process by using data mining and analytical tools to identify patterns or trends that may signal potential safety concerns.
When a potential safety signal is detected, a structured approach is used to assess its importance and accuracy. This stage requires a detailed review of all available data to determine its validity.
Key factors such as causality, patient population, and potential risk are meticulously considered, ensuring a comprehensive and systematic approach to pharmacovigilance.
Regulation to ensure the safety of GVP
Regulation, as we all know, is a vital aspect of every meaningful and good practice for human use.
The European Medicines Agency’s (EMA) good pharmacovigilance practices, released in 2012, is divided into 16 modules, each of which covers one major process in pharmacovigilance as a set of measures drawn up to facilitate the performance of GVP in the European Union (EU).
One of the major aspects is the requirement of establishing a pharmacovigilance risk assessment committee, also known as PRAC. It handles the risk management around the approval of a pharmaceutical product, so the market has maximized benefits. It covers signal detection, periodic safety update reports (PSURs), and more.
There are some post-approval reporting requirements like Periodic safety update reports (PSURs). It provides a comprehensive overview of the benefit-risk profile of a medicinal product. Signal management involves the detection, validation, and assessment of signals related to potential safety issues with medicinal products. Regulatory authorities may take enforcement actions, including product withdrawals, fines, or legal penalties if deficiencies in pharmacovigilance practices are identified.
Risk management plans (RMPs) outline the measures to be taken to identify, characterize, and minimize risks associated with a medicinal product. Pharmacovigilance inspections ensure compliance with pharmacovigilance regulations and guidelines. Inspections may focus on the systems, processes, and documentation related to pharmacovigilance activities.
Failure to comply with GVP requirements can have significant legal and financial consequences for pharmaceutical companies.
Regulation No 658/2007 posed by the European Commission also empowers the Commission to impose financial penalties on marketing authorization holders.
The idea behind this is to ensure the enforcement of certain obligations connected with marketing authorizations for medicinal products granted in accordance with Regulation (EC) No 726/2004.
The administrative penalties are usually fixed fines or based on company profits or levied daily.
The role of the marketing authorization holder (MAH)
The marketing authorization holder (MAH) plays a central role in the implementation of GVP and in safeguarding the ongoing safety of medicinal products throughout their lifecycle.
It should have specific quality system procedures and processes to ensure complement with legal requirements and guidelines. This system should cover the collection, evaluation, and reporting of adverse reactions and other safety-related information.
MAHs must appoint a qualified person for pharmacovigilance (QPPV) who is responsible for the overall pharmacovigilance activities. The QPPV serves as the primary contact point for regulatory authorities on these matters.
The marketing authorization holders are responsible for the development and implementation of risk management plans (RMPs) for certain medicinal products. RMPs outline strategies to identify, characterize, and minimize risks associated with a product.
They must provide a submission of periodic safety update reports (PSURs) to regulatory authorities at defined intervals. These reports provide a comprehensive overview of the benefit-risk profile of a medicinal product and contribute to the ongoing assessment of its safety.
They are obliged to report within 15 calendar days all suspected serious adverse drug reactions (ADRs) occurring within the EU to the Member States in whose territory the incident occurred.
MAHs have to submit Periodic Safety Update Reports (PSURs) to the Agency and the Member States, at least every six months during the first 2 years following authorization once a year for the subsequent two years, and at the time of application for the first renewal.
MAHs also play a critical role in communicating safety information to healthcare professionals, patients, and regulatory authorities. This includes the preparation and distribution of safety alerts, educational materials, and updates related to the use of their products.
GVP compliance and quality assurance
Quality assurance is a vital GVP necessity. That’s where a quality management system (QMS) is needed. It is a structured framework of documented processes, procedures, and responsibilities designed to achieve quality policies and objectives.
Through continuous monitoring and systematic feedback loops, a QMS facilitates ongoing improvement, making it an integral part of a successful, customer-focused organization.
Simultaneously, a robust Data Management System (DMS) is integral to pharmacovigilance.
These systems are built to collect, store, analyze, and report safety data—the core of pharmacovigilance. Ranging from basic databases to advanced software, they efficiently manage large amounts of information while ensuring data integrity and security.
Within this ecosystem, the QMS extends its reach to the software development aspect of the DMS. Here, the QMS ensures that the pharmacovigilance software solutions are developed and maintained to the highest quality standards, meeting not just the functional requirements but also complying with the rigorous regulatory standards.
It oversees the software development lifecycle, managing processes, minimizing errors, and ensuring that the software consistently aligns with the evolving needs of pharmacovigilance practices.
Also, companies need to establish training programs to ensure that employees are aware of their responsibilities and stay informed about changes in regulations and guidelines.
The training includes topics such as ensuring the safety of medicinal products, and the proper procedures for collecting, recording, and processing adverse event reports, including data entry and documentation.
These training programs may be conducted through various formats, including classroom sessions, online courses, workshops, and seminars. The goal is to ensure that personnel stay updated on evolving pharmacovigilance practices, regulatory requirements, and industry standards, contributing to the overall effectiveness of the pharmacovigilance system.
Future trends in GVP
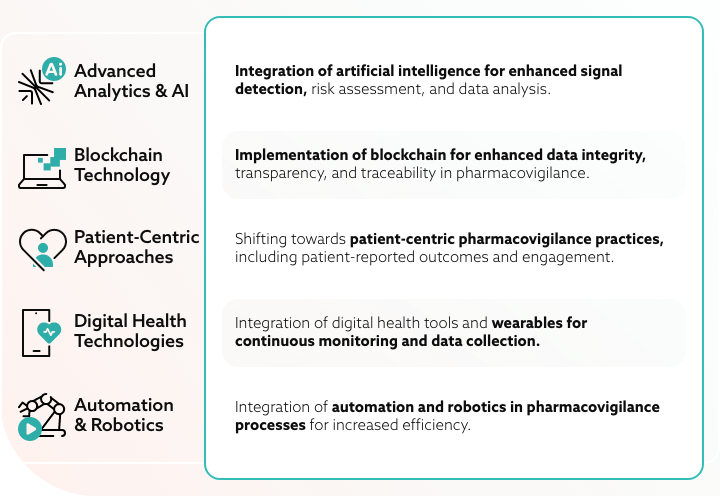
The dynamic field of pharmacovigilance practice should reflect the advancement of trends in technology, changes in regulatory expectations, and a constant improvement of patient safety.
Advanced healthcare analytics software, machine learning, and artificial intelligence (AI) are expected to play a significant role in signal detection, risk assessment, and data analysis.
In the realm of AI, for example, integrating advanced analytics and machine learning presents a paradigm shift in signal detection and risk assessment. Machine learning algorithms can adeptly analyze vast datasets, unveiling patterns and potential signals associated with adverse drug reactions.
Natural Language Processing (NLP) helps extract and analyze valuable information from unstructured text, such as medical literature and electronic health records. This enhances both the depth and efficiency of data analysis in pharmacovigilance.
A great example of AI being applied for pharmacovigilance is IBM Watson. It has been applied in drug safety to analyze and extract insights from large datasets, contributing to more efficient signal detection and risk assessment.
Simultaneously, Blockchain technology offers a strong solution for improving data integrity, transparency, and traceability in pharmacovigilance. It enhances security and creates a tamper-proof record of adverse events, ensuring reliable and trustworthy safety data.
Using chain-of-custody records within blockchain ensures a secure way to track the origin and handling of pharmacovigilance data. This is especially important when multiple stakeholders are involved in collecting, storing, and analyzing safety information.
Blockchain’s inherent capabilities facilitate secure and transparent data sharing among various entities involved in pharmacovigilance, fostering collaboration between pharmaceutical companies, regulatory authorities, and healthcare providers.
A direct case of blockchain being used in the industry is The MediLedger Project. It explores blockchain solutions for pharmaceutical supply chain integrity, which indirectly contributes to pharmacovigilance by ensuring the authenticity and traceability of medications.
This collective integration of AI and blockchain technologies not only streamlines processes but also establishes a robust foundation for the future of pharmacovigilance, marked by heightened efficiency and data security.
Staying alongside these future trends in GVP will reflect an ongoing commitment to improving patient safety and optimizing the risk-benefit balance of medicinal products.














