Healthcare technology

The Role of The Electronic Batch Record (EBR) Software In GMP
Company News

How Venture Builders are Transforming Digital Healthcare: Insights from Forbes Health Summit 2024

The Code of Change – The story of Ivaylo Ivanov and Ivan Lekushev from BGО Software

Ivan Lekushev – I am Dreaming Bulgaria to Become an IT Hub for Digital Healthcare
#TechTalks
BGO Software is Trusted by The World’s
Leading Healthcare organisations






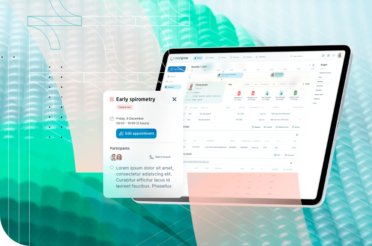
Elevate Your Hospital’s Clinical Trials
with a Flexible, Powerful CTMS
Easily migrate all current data. Customise to your unique needs.
Unlock seamless trial management.








 Ivo Kuzov
Ivo Kuzov
 Viktor Pavlov
Viktor Pavlov