Clinical trials serve as the cornerstone in the development of innovative treatments and improved methods for disease detection, diagnosis, and prevention. These trials, conducted on human subjects in controlled environments, provide vital insights into what works and what doesn’t in the realm of human health.
However, conducting clinical trials is a complex and costly process that demands precision, thoroughness, and efficient resource management. This is where clinical trial systems come into play, offering a solution to many of the challenges faced in clinical trials, from drug discovery to approval.
The market for clinical trials software is experiencing robust growth, driven by increasing demand from pharmaceutical organizations seeking to reduce costs, streamline operations, and gain access to advanced technologies.
The value of the global clinical trials management system market is expected to reach $4.72 billion by 2030, reflecting the importance and impact of these technological innovations in the life-sciences industry.(1)
To fully harness the potential of these innovative technology solutions, partnerships and collaborations are essential among all stakeholders involved in randomized controlled trials (RCTs).
This includes:
- researchers
- the biopharma and medical device industries
- clinical research organizations (CROs)
- health tech companies
- professional medical associations
- clinical health systems
- CMS private insurers (Centers for Medicare & Medicaid Services)
- regulatory authorities
Together, these players can drive forward the evolution and optimization of clinical trials, with profound benefits for medical research and, ultimately, patient care. Continue reading to find out everything you need to implement clinical trial software in your organization.
Types of e-clinical solutions you should know
E-clinical technologies encompass a range of tools designed to facilitate clinical research in various therapeutic areas. Different healthcare organizations and practices may utilize different combinations of these tools based on their specific needs.
In this article, we will start by exploring broadly applicable electronic systems, such as electronic health records (EHR), which are prevalent across various aspects of the healthcare sector.
Following this, we will transition into a detailed examination of more specialized software systems – those tailored specifically for managing clinical trials-showcasing how these focused technologies can significantly improve the efficiency and efficacy of clinical research processes.
Electronic health records (EHR)

In the last ten years, hospitals have seen a comprehensive shift from traditional paper-based medical records to electronic health records. EHRs are digital records updated in real-time, storing essential information such as patient diagnoses, administered treatments, lab tests, etc.
This change presents a remarkable opportunity for various aspects of clinical trials, including planning, design, initial site set-up, the actual execution of the study, data collection, assessing if the trial represents the target population appropriately, and following up on clinical events.
Electronic health records serve as a pivotal tool in determining participant eligibility for clinical trials. It’s essential to recognize that not all individuals qualify for every clinical trial, as there are specific guidelines known as “inclusion and exclusion criteria”.
These criteria may encompass a range of factors such as age, gender, the type and stage of the disease, and the presence of other medical conditions. EHRs can help estimate how these rules affect the number of people you can recruit for the trial at different locations.
EHRs can help identify locations like hospitals or healthcare technology platforms that have many potential trial participants. Electronic health records can also give information about how often certain health events (like heart attacks, strokes, etc.) occur in real-world settings. This can help you decide how many people you need in your trial and how to do the statistical tests to analyze your data.
Overall, EHRs are changing the way clinical trials are planned and carried out. For example, a tool called the InSite platform, used in many hospitals across Europe, can look at EHR data to find out how many patients might be eligible for a particular trial and how often certain health events occur in these groups. This information is then used to plan the trial more effectively.
However, to fully leverage the potential of EHRs and bring about a revolution in the conduct of trial operations, it’s crucial to tackle several substantial hurdles through collaborative efforts involving multiple stakeholders.
These challenges include ensuring the accuracy and completeness of EHR data, addressing privacy concerns related to patient data, and overcoming issues related to platform interoperability. By overcoming these obstacles, EHRs could truly reshape the landscape of clinical trial operations, making them more efficient and effective.
Digital health applications, biosensors and wearables
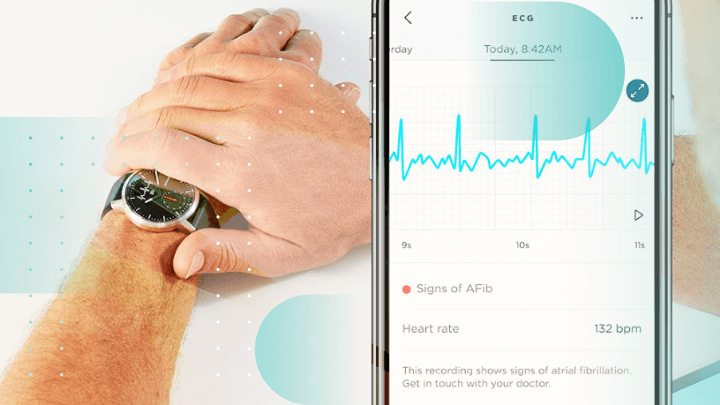
The majority of US residents and over half of the global population own a smartphone, a figure that continues to grow. Mobile health (mHealth) apps can shift many clinical trial activities to patients’ locations, including enrollment and real-time data collection.
Biosensors and wearables, enabled by mHealth, can monitor participants’ vitals and daily activities, generating data for digital biomarkers like sleep quality and heart rate.
However, technical and operational issues, along with the need to prove reliability, pose challenges to the widespread use of mobile and biosensor data in trials. Furthermore, not everyone has access to smartphones or wearables, potentially excluding certain patient groups.
To avoid this, alternatives for more inclusive patient recruitment and participation should be considered, like internet participation or traditional study methods.
Specific clinical trial solutions
Healthcare organizations can significantly streamline their clinical trial processes through a unified management platform encompassing various modules.
However, each module can also function as an independent solution in a specific therapeutic area, tailored to specific needs:
- Clinical trial management system (CTMS). This module provides centralized management for all operational aspects of clinical trials, including planning, tracking, and reporting, improving efficiency and compliance.
- Electronic trial master file (eTMF). This digital module replaces traditional paper files, storing, managing, and tracking all essential clinical trial documents, thereby ensuring regulatory compliance and easy access.
- Electronic data capture (EDC). This module facilitates the collection, storage, and management of trial data in an electronic format, improving data accuracy, and speeding up data analysis.
- Randomization. This module helps in random assignment of trial participants to treatment groups, a critical component in reducing bias and ensuring the validity of trial results.
- Safety and pharmacovigilance. This module aids in collecting, analyzing, and responding to any adverse events or drug reactions during the trials of new drugs, ensuring participant safety and regulatory compliance.
- Budget management. This module provides tools for budgeting, cost tracking, and financial reporting for the trial, enabling efficient allocation of resources and financial control.
Together, these modules create a comprehensive toolset to manage all facets of a clinical trial, providing a cohesive yet flexible solution for all stakeholders involved in RCTs. Now we will move on to exploring some of the modules that can serve as a separate software solution for healthcare organizations.
Electronic data capture (EDC) systems
Electronic data capture software is a digital tool used in clinical trials to collect, store, and manage data in an electronic format. It’s a key technology that has revolutionized the way data is collected, replacing traditional paper-based data collection methods.
EDC systems are designed to streamline and automate data collection processes, providing a more efficient and accurate way to gather and analyze clinical trial data. For example, in the SAFE-PCI for Women trial, an electronic data capture system autopopulated baseline characteristics, reducing the workload for study coordinators by 65%.
The SWEDEHEART national registry used a similar approach to collect data in a series of transformative trials at a much lower cost than traditional methods.
Clinical trial management systems (CTMS)
A clinical trial management system (CTMS) is an essential software platform that orchestrates all facets of a clinical trial, thereby serving as a cornerstone for realizing operational efficiencies.
It centralizes the administrative components of each study, including planning, reporting, and tracking:
- Trial planning and design. CTMS software aids in outlining the study’s structure and workflow, assisting in innovative study designs, protocol development, and determining budget and resources required.
- Study conduct. It helps manage various aspects of a study such as patient recruitment, tracking patient progress, site monitoring, and ensuring protocol adherence.
- Data management and reporting. A CTMS provides tools for data collection, management, and statistical analysis. It generates various reports based on advanced analytics, allowing research teams to monitor trial progress and make data-driven decisions.
- Project management. The software enables tracking of tasks, deadlines, and milestones. It also helps in risk-based monitoring and managing the team’s workload and assignments, promoting efficient collaboration among team members.
Randomization & trial supply management (RTSM) software
Randomization and Trial Supply Management (RTSM) is a technology used in clinical trials to manage two key aspects: patient randomization and supply chain management.
Randomization
In a clinical trial, randomization is the process of randomly allocating participants into different study groups, such as the control group and various treatment groups. This is a crucial step in reducing bias and ensuring the results are statistically valid.
The process can involve complex randomization strategies, such as stratified or block randomization, to maintain balance across treatment groups and consider factors like disease severity or demographic variables. RTSM systems are used to handle this complex process efficiently and accurately.
Trial supply management
Clinical trials involve the distribution of investigational drugs or medical devices to multiple research sites, often in different locations or countries. Managing this supply chain can be complex, particularly in trials that are blinded (where the investigators, and sometimes the participants, do not know which treatment group they are in).
RTSM is used to manage the inventory and distribution of trial supplies, ensuring that each trial site has enough of the necessary supplies, that the supplies are used before their expiry date, and that the blinding of the trial is maintained.
Why should stakeholders implement clinical trial software?
Cost efficiency
EHR-based or mHealth-enabled clinical trials have shown potential for substantial cost reduction. For instance, the ADAPTABLE trial, with 20,000 patients, would have likely cost over $150 million if conducted traditionally, but due to the implementation of EHR and mobile technologies, its budget was almost a tenth of that.
However, achieving such savings demands significant infrastructure. The EHR4CR project, for example, involved 34 partners and ran on a budget of about $18 million from 2011 to 2015 to create an EHR platform across a large network. Setting up scalable and sustainable infrastructure for these types of trials requires a long-term commitment from funders and health systems.
Optimized trial processes
Clinical trial software plays a pivotal role in optimizing trial processes, thus leading to better trial performance. It streamlines a multitude of tasks such as patient recruitment, trial planning, data collection, and analysis.
These tools automate repetitive tasks, which are usually time-consuming, and allow researchers to focus more on the substantive aspects of the trial. It also fosters a seamless workflow, providing quicker access to clinical research data and accelerating the decision-making process.
Improved data accuracy
One of the crucial advantages of clinical trial software is improved data accuracy, which has a direct effect on patient outcomes. With electronic data capture (EDC) capabilities, it eliminates the risk of errors associated with manual data entry. It ensures data integrity and quality, thereby enhancing the credibility of the trial results, and improving patient outcomes.
Enhanced collaboration among stakeholders
Clinical trial software fosters seamless collaboration among various stakeholders involved in a clinical trial, including researchers, clinicians, sponsors, and patients. It provides a centralized digital platform where all parties can communicate, share relevant information, and work collaboratively towards common goals. It ensures that everyone stays updated and aligned, promoting efficiency and transparency in trial conduct.
Reduced oversight and human error
Clinical trial systems significantly reduce the potential for oversight and human error. The automated nature of these tools minimizes the chance of missing crucial clinical data or tasks.
Additionally, built-in safety and compliance checks ensure adherence to regulatory standards and protocols. This leads to increased overall quality of trials and mitigates the risks of costly mistakes.
Challenges and considerations in implementing clinical trial software
Implementing clinical trial software presents various challenges and considerations, which can be broadly categorized into technical, organizational, and regulatory aspects:
Technical challenges
The successful implementation of clinical trial software hinges on several key considerations. Foremost among these are data integration and interoperability. In the complex landscape of clinical trials, different systems often utilize distinct data formats and standards, which can pose significant challenges to seamless and powerful integration.
Therefore, it is crucial that the chosen software be designed to communicate effectively and share data seamlessly with these diverse systems, facilitating fluid inter-system interaction.
Moreover, considering the sensitive nature of the data involved in clinical trials, data security is another paramount concern. Robust cybersecurity measures must be in place to protect this data from potential breaches.
These measures should include, but not be limited to, data encryption, regular security audits, and strict adherence to regulatory standards like the Health Insurance Portability and Accountability Act (HIPAA).
Finally, the scalability of the system is a key factor that cannot be overlooked. As clinical trials progress, they often generate increasing volumes of data. Additionally, there may be instances where multiple trials are being conducted concurrently.
In these scenarios, the software system must be capable of handling this surge in data volume without compromising on its performance or functionality. This scalability allows the system to adapt to the evolving needs of clinical trials, ensuring that it remains a valuable tool throughout the trial process.
Organizational challenges
Implementing a new software system in the clinical trial process can pose challenges. The cost of the system is a significant factor, encompassing not only the purchase price but also expenses related to installation, maintenance, training, and potential future upgrades.
Additionally, comprehensive staff training is crucial to ensure smooth operation, though it may temporarily impact productivity. Finally, effective change management is vital to overcome resistance. This can be achieved by clearly communicating the system’s benefits and involving end-users in planning and decision-making, fostering a collaborative approach to implementation.
Successful implementation of clinical trial software solutions
Successfully implementing clinical trial software involves several steps, and there are important considerations that all stakeholders need to be aware of:
- Needs assessment. First, your organization should identify its specific needs and define the objectives the software should help achieve. These requirements could range from data management, patient recruitment, study design, to regulatory compliance, etc.
- Research and vendor selection. Next, conduct thorough research on available software options. Consider factors such as the software’s functionality, user-friendliness, cost, customer support, security features, and scalability. Ask for demos or trial periods to evaluate if the software meets your needs.
- Data migration. Plan for a smooth transition from your existing system to the new one. This could involve data mapping, data cleaning, or migration testing.
- Testing and validation Before going live, the system should be thoroughly tested to ensure it works as expected. This includes functional testing, user acceptance testing, and software validation.
- Post-implementation review and support. After implementation, regularly review the system’s performance. Vendor support should be available for troubleshooting and system updates.
Remember, communication and change management are key. Ensure all stakeholders understand why the new software is needed, how it will improve their work, and how to use it. This will reduce resistance and ensure a smoother transition.
Future trends and advancements in clinical trial software development
Artificial intelligence and machine learning

AI and machine learning have the potential to transform various sectors, including the conduct of clinical trials. They can enhance patient recruitment by analyzing digital data, enabling targeted marketing strategies. Predictive models can also help identify eligible participants by analyzing various data sources like electronic health records, lab results, and vital signs.
Applying machine learning to clinical trial data can help identify trends or issues more quickly, reducing the need for on-site monitoring and boosting trial efficiency and quality. Digital behavior patterns can pinpoint participants who may need more support to stick to the study treatment, helping allocate resources effectively.
AI could also automate trial endpoint decisions, improving efficiency and reducing costs. However, integrating these digital solutions into clinical trials requires further development.
Virtual trials

Virtual trials, also known as decentralized trials, leverage cutting-edge digital technologies to conduct certain aspects of the trial remotely, reducing the need for physical presence at the clinical sites.
These trials incorporate elements like remote monitoring, which uses digital tools to track health indicators, telemedicine visits, which allow for virtual check-ins and consultations, and digital consent, which streamlines the process of obtaining and managing participant consent.
The advantages of virtual trials are manifold. They can significantly improve patient access by eliminating geographical barriers and making participation more convenient, particularly for those with mobility issues or those residing in remote areas.
Furthermore, by making the trial process less burdensome for participants, virtual trials may reduce drop-out rates, thus maintaining the integrity and statistical power of the trial.
Despite the advantages of virtual trials, they may pose challenges in terms of data privacy and security, technology access and literacy among participants, and regulatory compliance, which necessitates additional development to address these issues effectively.
In conclusion, harnessing the power of innovative software solutions is key to streamlining clinical trials in today’s fast-paced, data-driven world. These innovative technologies offer the potential to transform the clinical trial landscape, making it more efficient, cost-effective, and patient-centered.
Whether it’s EHRs, EDC tools, or the burgeoning realm of virtual trials, the future of clinical trials is digital. However, implementing these systems requires expertise and experience. If you’re looking to integrate these innovative solutions into your clinical trial processes, don’t hesitate to reach out to BGO Software, a trusted partner in the clinical research industry.
References:
The global clinical trials management system market size is anticipated to reach USD 4.72 billion by 2030, expanding at a CAGR of 14.3% from 2023 to 2030. (1)
Sources:
- Cloudbyz | CTMS | CTBM | eTMF | EDC | PPM: https://www.cloudbyz.com/
- Technology-Enabled Clinical Trials | Circulation – AHA Journals: https://www.ahajournals.org/doi/10.1161/CIRCULATIONAHA.119.040798
- https://blog.formedix.com/how-clinical-trial-software-can-be-used-to-optimize-clinical-trials














